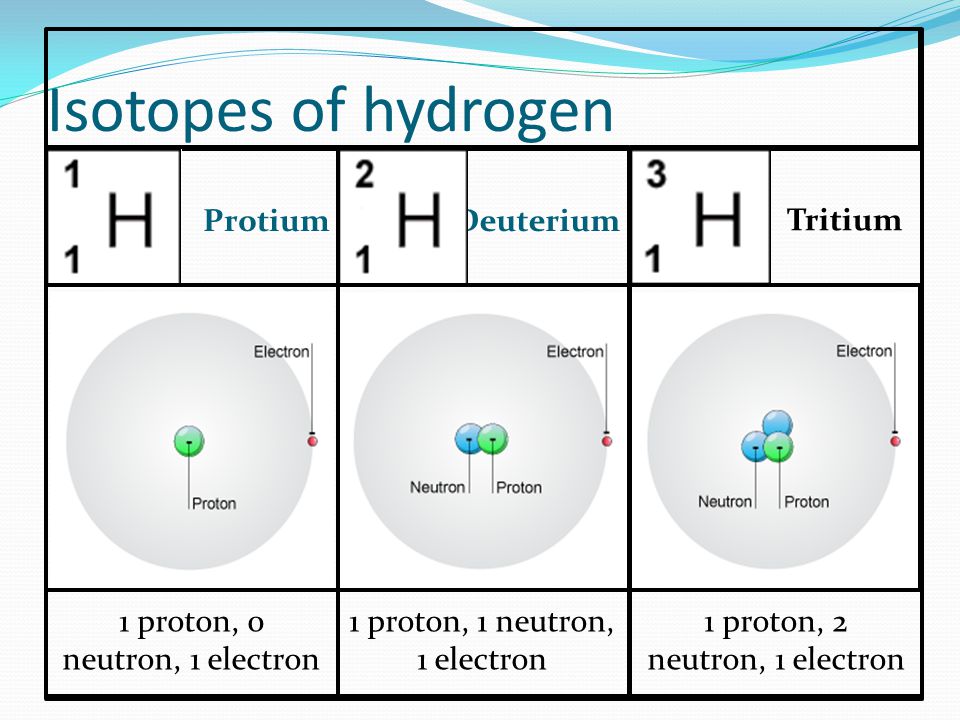
Key thing to remember: The proton number is what defines an element, if that changes it becomes a different element! Isotopes and Relative Atomic Mass. - ppt download

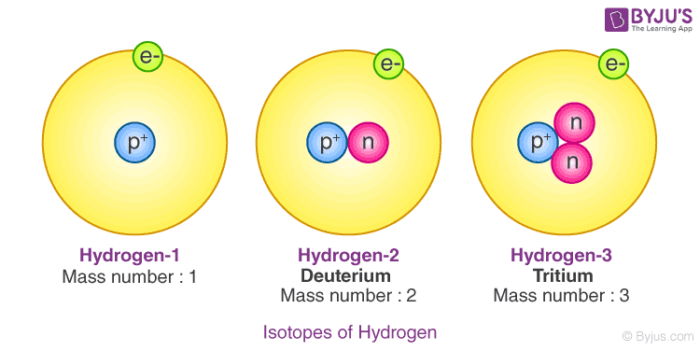
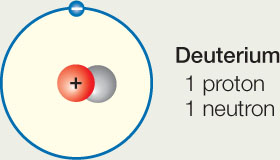
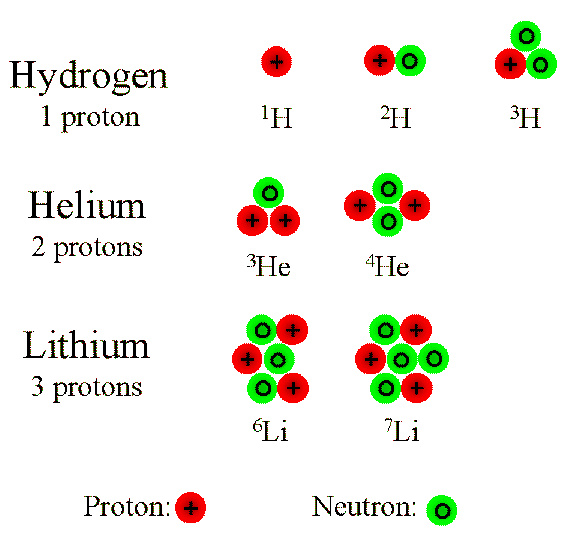
Hydrogen-1 Contains 1 proton and 1 electron – as all isotopes of H must contain. No neutrons The most common type of H atom. - ppt download
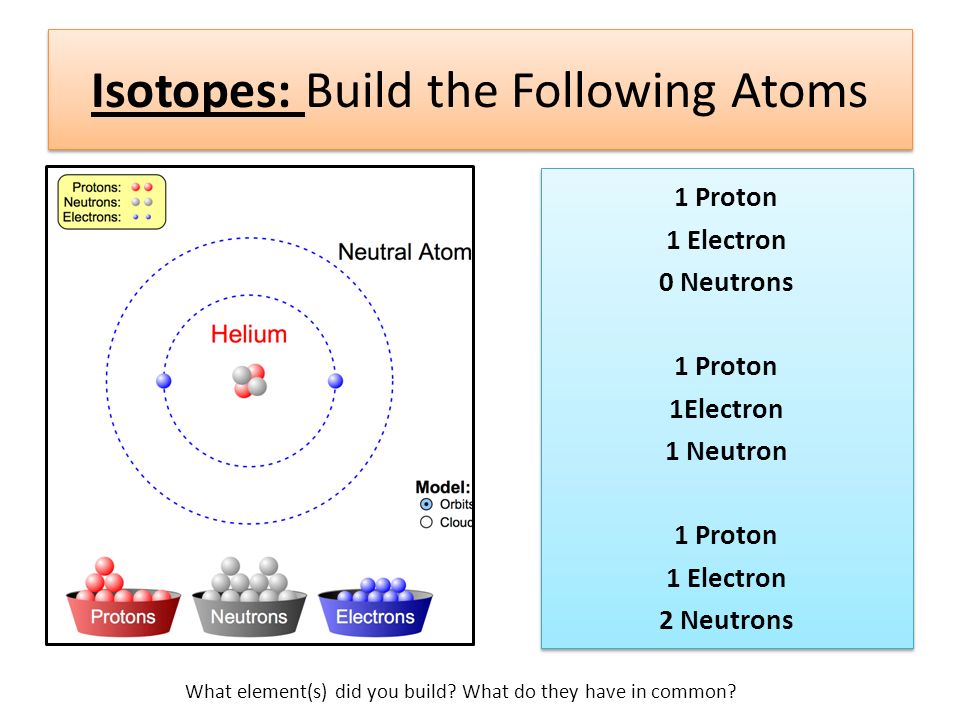
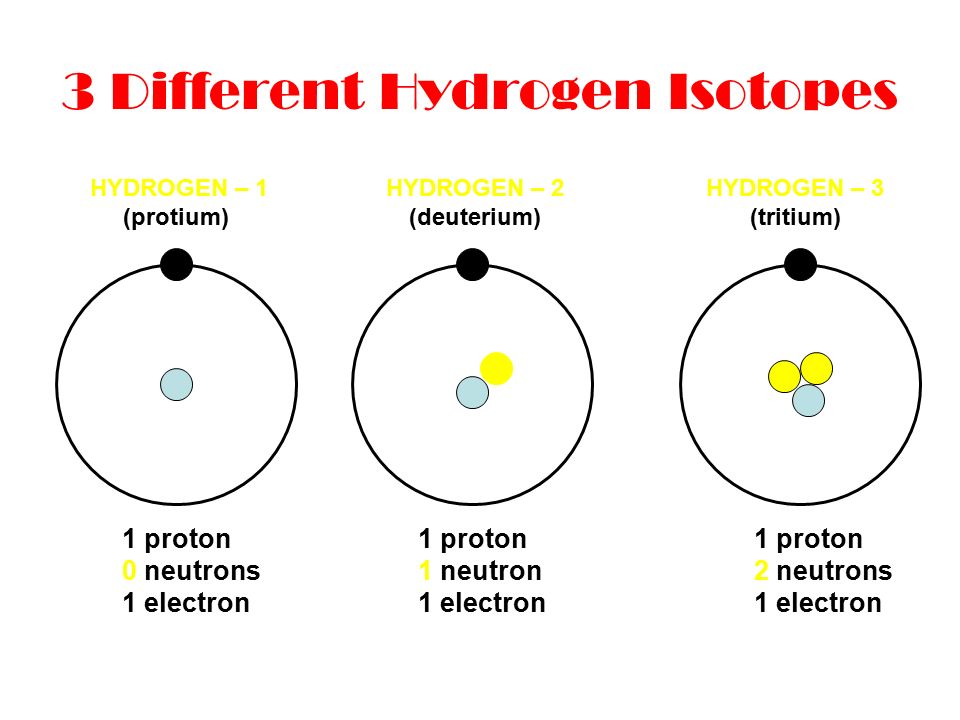
Isotopes: Build the Following Atoms 1 Proton 1 Electron 0 Neutrons 1 Proton 1Electron 1 Neutron 1 Proton 1 Electron 2 Neutrons 1 Proton 1 Electron 0 Neutrons. - ppt download

The Proton and its quarks. b. The neutron consists of two down-quarks... | Download Scientific Diagram

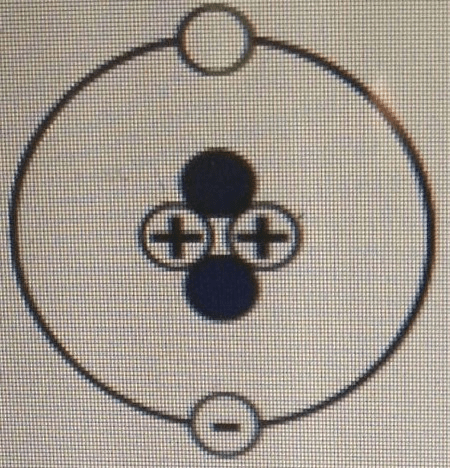
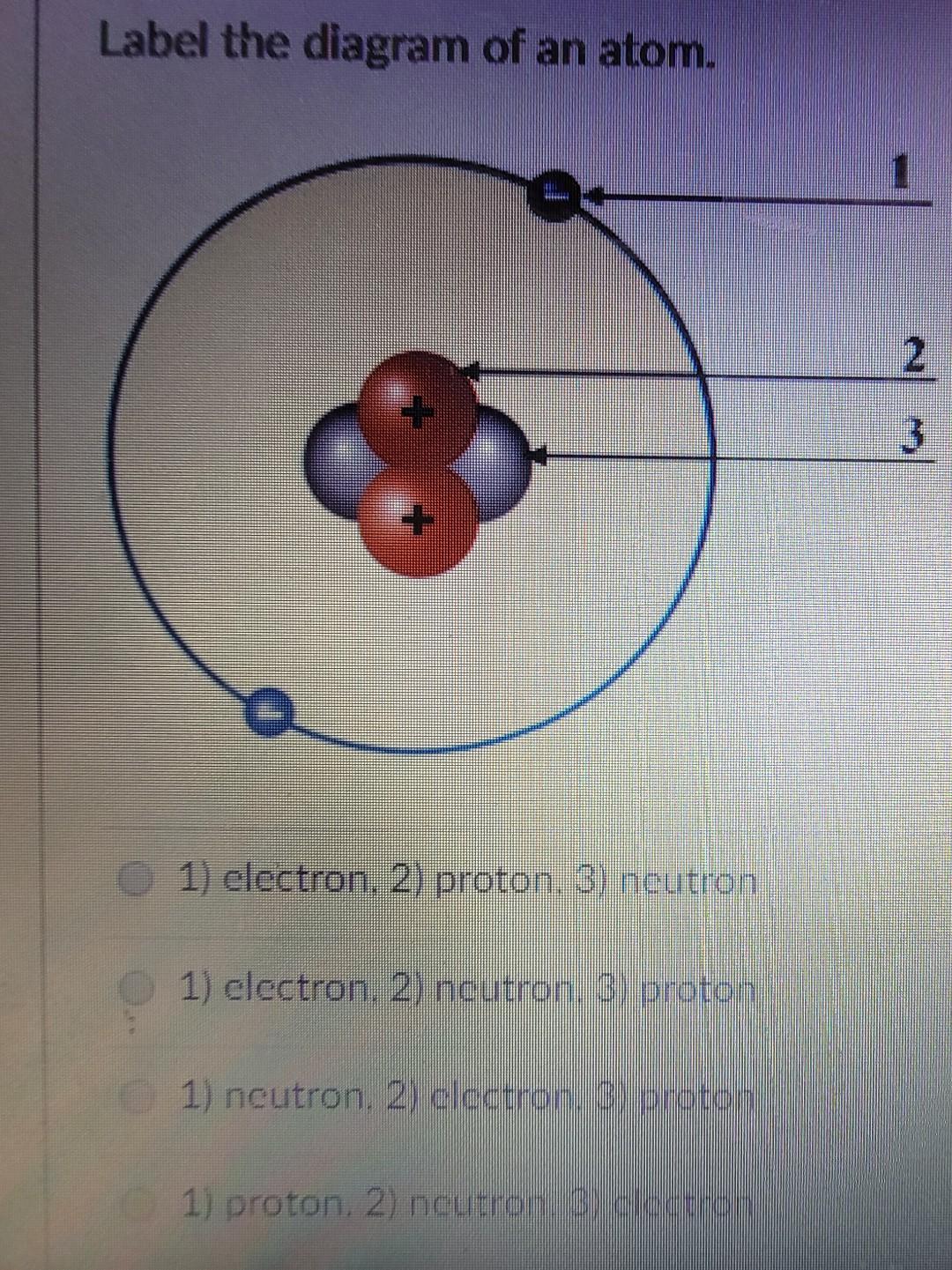
Which set of labels should be used to correctly complete the atomic model below? a. 1 - Electron 2 - - brainly.com
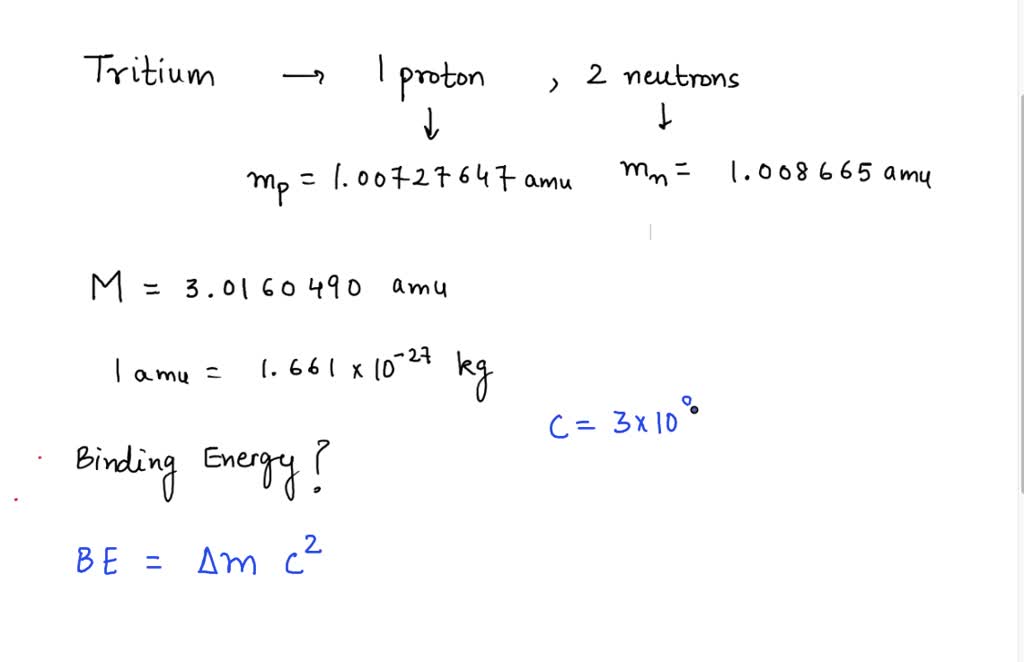
SOLVED: Tritium is an isotope of hydrogen that has 1 proton and 2 neutrons. What is the binding energy of a tritium nucleus if it has a mass of 3.0160490 amu? (Hint: